The U.S. Food and Drug Administration (FDA) recently announced that pharmacies can now distribute abortion drugs for the first time.
Bishop Michael F. Burbidge of Arlington, Virginia, chair of the U.S. Catholic bishops’ Committee on Pro-Life Activities, spoke out against this decision, stating that “The FDA should protect the life and health of both mothers and children, not loosen safety standards under industry or political pressures.”
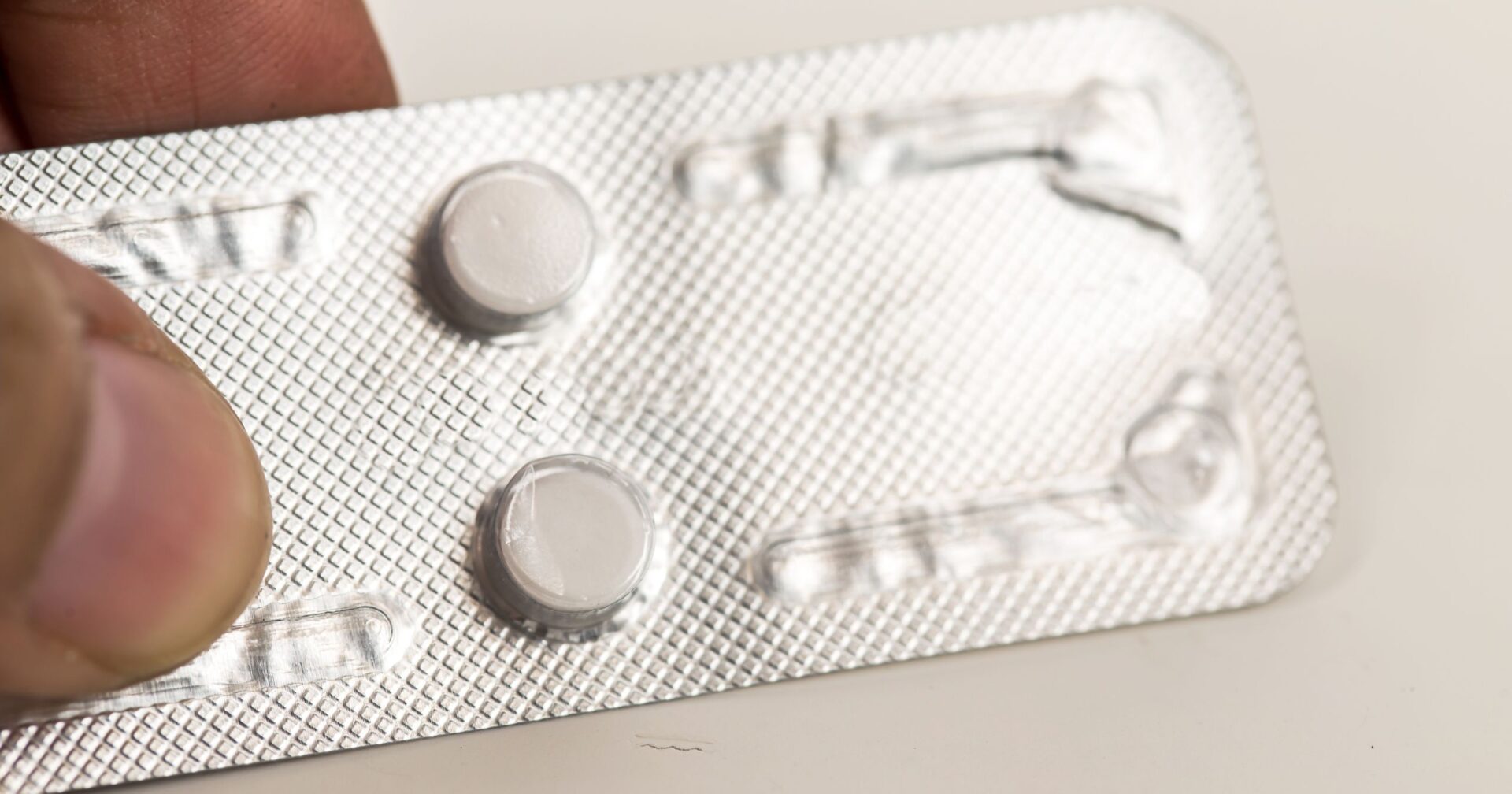
Mifepristone, which is paired with another drug called misoprostol, is taken by pill and approved by the FDA for use up to 10 weeks of gestation. It accounts for more than half of all abortions in the U.S., according to the Guttmacher Institute.
Burbidge cited the Charlotte Lozier Institute’s fact sheet on the risks and complications of abortion by pill, stating that “The rate of serious complications after chemical abortion is considerably higher than after surgical abortion.”
He also warned that this decision “facilitates the isolation of critically vulnerable pregnant women and invites more risk, pain, and trauma” and may result in violations of conscience for pharmacy workers who cannot dispense such drugs.
In December 2021, the FDA lifted restrictions on mifepristone distribution, allowing doctors to prescribe the drugs online and mail the pills to patients, enabling pregnant women to undergo early abortions without visiting a medical facility in person.
Bishop Burbidge reaffirmed the Catholic Church’s position on life, saying:
“The Catholic Church is consistent in its teaching on upholding the dignity of all life, and that must include care for both women and their children. We decry the continuing push for the destruction of innocent human lives and the loosening of vital safety standards for vulnerable women.”
According to CNA, the FDA did not provide comment on the matter.
Pray for life!
🙏